Dental X-Ray Equipment Infection Control: Essential Sterilization and Safety Protocols

Proper infection control and sterilization procedures for dental X-ray equipment are essential for preventing cross-contamination and maintaining patient safety. With increasing regulatory scrutiny and evolving infection control standards, dental practices must implement comprehensive protocols that address all aspects of radiographic equipment sterilization.
Understanding the Critical Points of Contamination
Digital X-ray sensors, positioning devices, and associated equipment present multiple opportunities for cross-contamination during patient care. Unlike traditional film-based systems, digital sensors require specialized handling to maintain both sterility and equipment functionality.
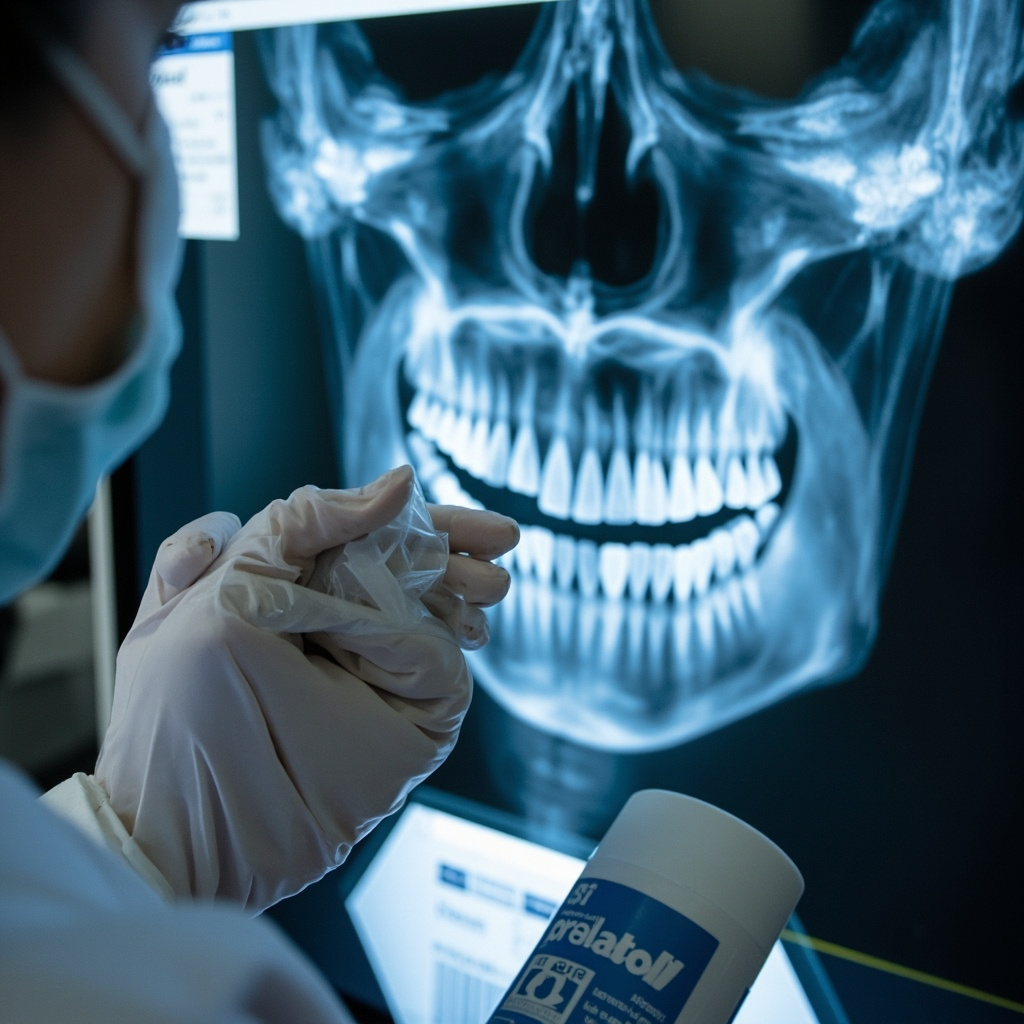
The sensor surface, connector cables, and positioning aids all require attention during the sterilization process. Failure to properly address these contact points can result in pathogen transmission between patients and potential regulatory violations.
CDC Guidelines for Digital Radiography Equipment
The Centers for Disease Control and Prevention provides specific guidance for dental radiographic equipment sterilization:
- Heat-sterilizable accessories: All bite blocks, positioning rings, and guides that can withstand autoclave temperatures should undergo routine steam sterilization
- Digital sensor protection: FDA-cleared barrier envelopes must protect sensors during use, with additional high-level disinfection between patients
- Surface disinfection: Equipment surfaces require EPA-registered intermediate-level disinfectants with appropriate contact times
- Cable management: Sensor cables should be covered with protective sleeves or undergo surface disinfection protocols
Barrier Protection Systems
Implementing comprehensive barrier protection significantly reduces contamination risk and simplifies sterilization workflows. Modern barrier systems include sensor-specific sleeves, cable covers, and positioning device protectors.
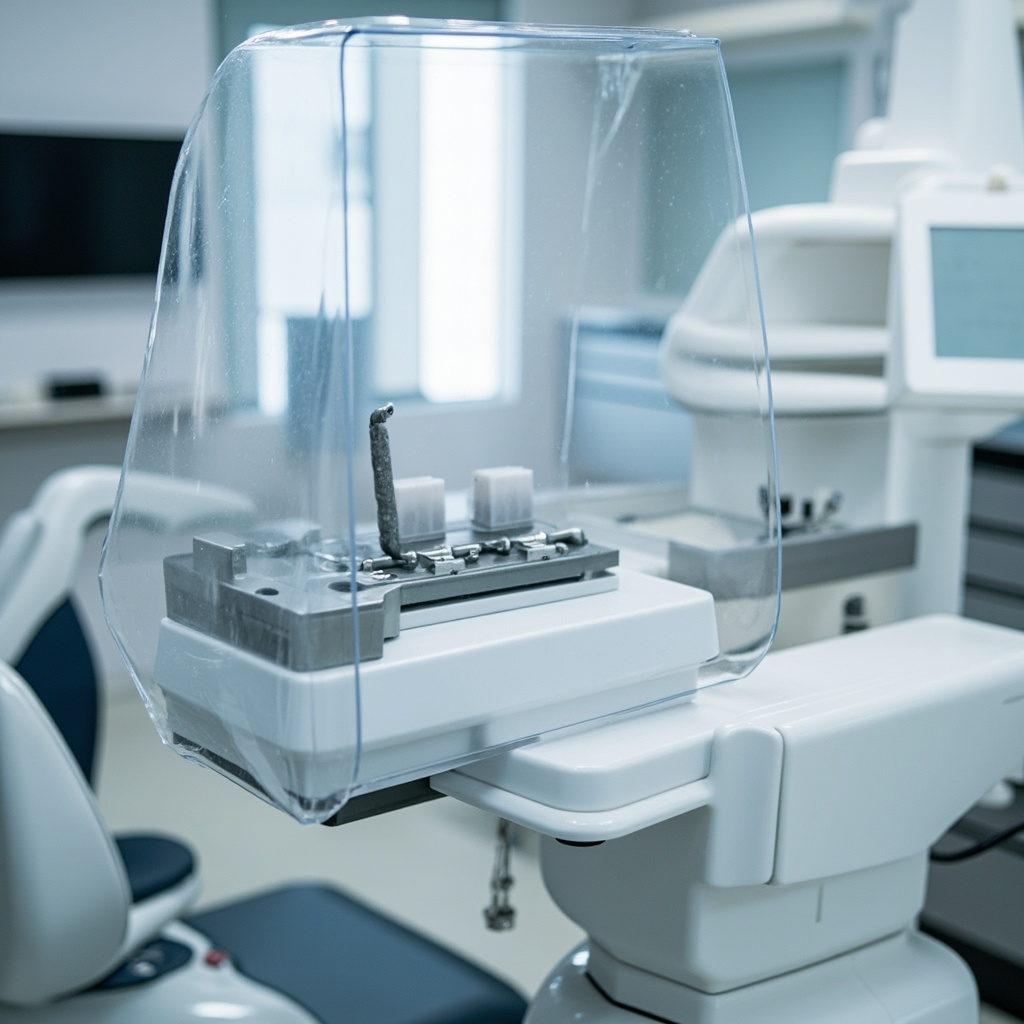
Quality barrier materials must be puncture-resistant, moisture-proof, and designed for single-use applications. The barrier envelope should completely encase the sensor while maintaining image quality and patient comfort during radiographic procedures.
Sterilization Protocols for Heat-Sensitive Equipment
Digital sensors and electronic components cannot undergo traditional autoclave sterilization. These items require high-level disinfection using EPA-approved chemical agents specifically cleared for semi-critical dental devices.
Chemical Disinfection Procedure:
- Pre-clean all visible debris using appropriate cleaning solutions
- Apply EPA-registered high-level disinfectant for manufacturer-specified contact time
- Ensure complete surface coverage without pooling or excess moisture
- Allow proper air-drying before storage or next patient use
- Document disinfection cycles for regulatory compliance
Staff Training and Compliance Monitoring
Effective infection control requires comprehensive staff training on equipment-specific protocols. Team members must understand the distinction between sterilization and disinfection, proper barrier application techniques, and documentation requirements.
Regular compliance audits should verify adherence to established protocols, proper chemical concentrations, and appropriate contact times. Many practices implement checklists and monitoring systems to ensure consistent application of sterilization procedures.
Regulatory Considerations and Documentation
State dental boards and accreditation organizations increasingly scrutinize infection control practices during inspections. Practices must maintain detailed records of sterilization cycles, chemical testing logs, and staff training documentation.
Current regulatory trends emphasize evidence-based protocols, regular process validation, and continuous quality improvement in infection control practices. Failure to maintain proper documentation can result in significant regulatory penalties and patient safety risks.
Cost-Effective Implementation Strategies
While comprehensive infection control requires investment in barrier materials and chemical disinfectants, the long-term benefits include reduced equipment contamination, extended sensor lifespan, and enhanced patient safety. Many practices find that bulk purchasing of barrier supplies and standardized protocols reduce overall costs while improving compliance.
Implementing automated monitoring systems and staff training programs creates sustainable infection control workflows that adapt to evolving regulatory requirements and emerging pathogens.
Future Considerations
Emerging technologies in antimicrobial coatings, UV sterilization systems, and automated disinfection equipment may reshape dental radiography infection control. Practices should stay informed about advancing technologies while maintaining current compliance with established CDC guidelines and manufacturer recommendations.
Regular review and updating of infection control protocols ensures continued patient safety and regulatory compliance in an evolving healthcare environment.